

【AACR 2025】上细前沿冲破五 | AI驱动免疫医治药物快速开发:CARP智能平台实现VHH高通量精准筛选 2025-05-15 4月25日-30日,备受存眷的美国癌症研究协会(AACR)年会于美国芝加哥召开,本届集会以 同一癌症科学与医学:连续立异带来厘革 为主题。秉持 疗效为核,黎民可和 的药物开发理念,上海细胞医治集团连续鞭策JL-闪CAR-T 为焦点的要害技能平台开发,实现原创非病毒载体基因写入技能、纳米抗体VHHMAb 技能等底层技能的连续冲破,加快立异医治药物开发进程。本次集会,集团共有5项最新研究结果获邀现场展示,笼罩从底层平台冲破到上风抗体发明,再来临床运用转化的完备药物开发系统。充实揭示了集团于改造CAR-T等立异细胞药物可和性及治愈性方面的前瞻结构和广漠远景。 针对于传统CAR-T抗体发明的瓶颈,CARP平台(CAR-T AI-powered Rapid high-throughput screening Platform)为首个AI驱动、全谱系高通量的纳米抗体(VHH)筛选解决方案。由上海细胞医治集团美国子公司Chantibody(CHANTI三木SEO-BODY THERAPEUTICS INC.)研发搭建。 上海细胞医治集团与其美国子公司Chantibody(CHANTIBODY THERAPEUTICS INC.)互助开发的CARP平台,可以或许显著加快新型免疫疗法的发明与临床前评估,为CAR-T疗法的成长提供了精准、可扩大的新路径。将来,将进一步开展体内疗效及安全性验证,并拓展至更多肿瘤抗原及自身免疫疾病靶点。 择要号:5889 择要标题问题: CARP: A High-Throughput Platform for Rapid VHH Discovery of Potent CAR-T Therapy 择要内容: 传统CAR-T筛选包括:先筛选特异性联合方针抗原的抗体,再将抗体作为胞外抗原辨认区制备嵌合抗原受体,然后体外验证效果。然而,传统CAR-T筛选面对显著的瓶颈问题:1. 抗体筛选速率慢,2. 难以高通量辨认具备多样表位笼罩的、高质量抗体, 3. 往往因表位笼罩不全致使功效漏检或者误选。这些因素配合致使了CAR-T研发周期延伸,资源耗损加重,而且可能错掉真实的具备临床价值的份子。 VHH以其份子量小、可模块化拼接的上风,为构建双/多特异性CAR-T提供了抱负支撑,但要实现高通量、全谱系的功效化筛选,仍需新一代技能平台。咱们基在CAR-T筛选的痛点搭建了CARP高通量筛选平台。CARP平台于短期内有用辨认并聚焦潜于高亲及力、高特异性VHH。本次研究基在CARP对于CDH17(Cadherin-17)卵白免疫羊驼与美洲驼所患上的VHH举行高通量筛选,加快得到了优化的VHH CAR-T(图1)。 于完成VHHMAb 技能平台CDH17 VHH谱系阐发而且建库后,得到 400具有优良的人源化,免疫原性的VHH。并举行了多细胞模子共造就验证CART的方针依靠性的增殖能力。于高表达CDH17的H716细胞、中等表达的Colo205细胞与CDH17阴性的HCT-116细胞中,CAR-T的扩增体现出显著差异。H716共造就组CAR-T阳性率和倍增幅度最高,Colo205组中等,HCT-116组则基本无增殖(图2)。该成果直不雅证实了候选VHH于差别表达程度肿瘤细胞中的方针依靠性扩增能力。 Figure 2. The preparation of pooled CAR-T and serial proliferation by co-culturing with target+ cell lines. After CAR-T preparation (A), the percentage (B) and fold change (C) of CAR-T proliferation were assessed after co-culturing with H716 (CDH17 high), Colo205 (CDH17 medium), and HCT-116 (CDH17 negative) cells. NGS测序和增殖能力排序,经由过程对于筛选池中每一轮共造就后细胞群体举行NGS测序,获取各克隆的相对于品貌变化曲线,并用斜率模子举行拟合(图3A、3B),实现对于数百候选的快速定量排名;随后使用层级聚类(图3C)分层筛选出高潜力(快速斜率上升)与低潜力(曲线平缓或者降落)VHH群体。 经由过程验证筛选成果发明CARP具有高正确率。从NGS和斜率排序中遴选20个高潜力克隆及6个低潜力克隆举行功效验证。高潜力组:19/20个克隆于H716共造就中体现出强劲扩增(图4A);低潜力组:5/6个克隆险些无扩增(图4B);传统筛选比照:仅2/5个通例亲及力优选克隆体现出优良扩增(图4C)。该验证系统精准区别出功效强弱,证实CARP于候选VHH筛选上的高效与靠得住。 Figure 4. Leads validation to confirm CARP accuracy in lead selection. Selected CART leads were co-culturing with H716 cells. (A) In the group of positive clones, 19 out of 20 demonstrated strong CAR-T expansion. (B) In the group of negative clones, 5 out of 6 showed no or minimal CART expansion. (C) For comparison, conventional CAR-T leads (selected based on good binding affinity to proteins and cells) were tested, and only 2 out of 5 exhibited strong CAR-T expansion. 结论: 1. 高正确率掷中:20个功效阳性克隆中有19个于后续功效试验中一致体现优秀;6个阴性克隆中5个未能增殖,验证了CARP平台的筛选精度。 2. 优质VHH候选:乐成辨认多个靶向CDH17统一布局域的VHH, CAR-T的扩增与肿瘤细胞杀伤活性均优在临床比照与传统筛选份子。 3. 加快开发路径:平台一次免疫、一次筛选便可完成人源化及功效评估,年夜幅缩短新型CAR-T疗法的前期研发周期。


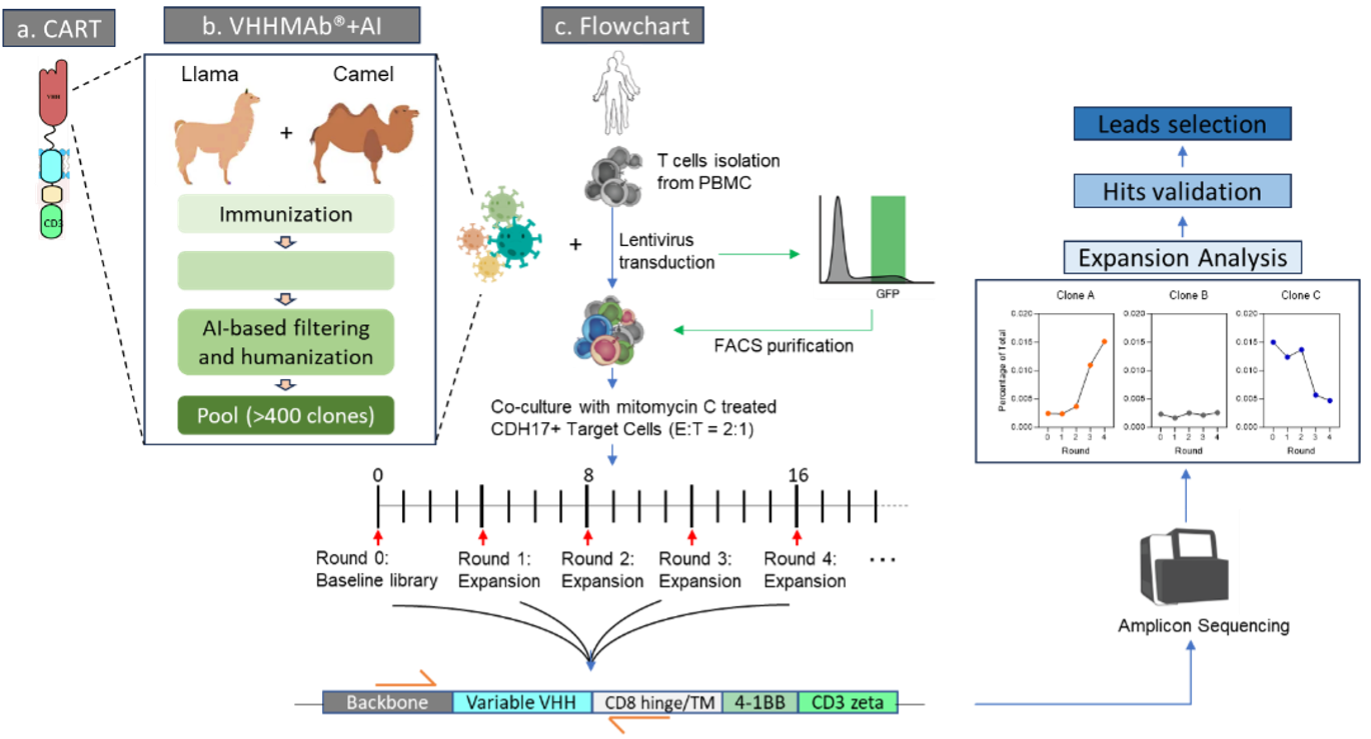 Figure 1. CARP high throughput screening workflow: It begins hits discovery by using VHHMAb platform, followed by AI-powered VHH library construction that encompasses antibodies with diverse epitopes and low i妹妹unogenicity. This is followed by pooling CAR-T clones, co-culturing with target cells, performing NGS of amplicons from the amplified CAR-T clones, and concluding with lead validation.
Figure 1. CARP high throughput screening workflow: It begins hits discovery by using VHHMAb platform, followed by AI-powered VHH library construction that encompasses antibodies with diverse epitopes and low i妹妹unogenicity. This is followed by pooling CAR-T clones, co-culturing with target cells, performing NGS of amplicons from the amplified CAR-T clones, and concluding with lead validation.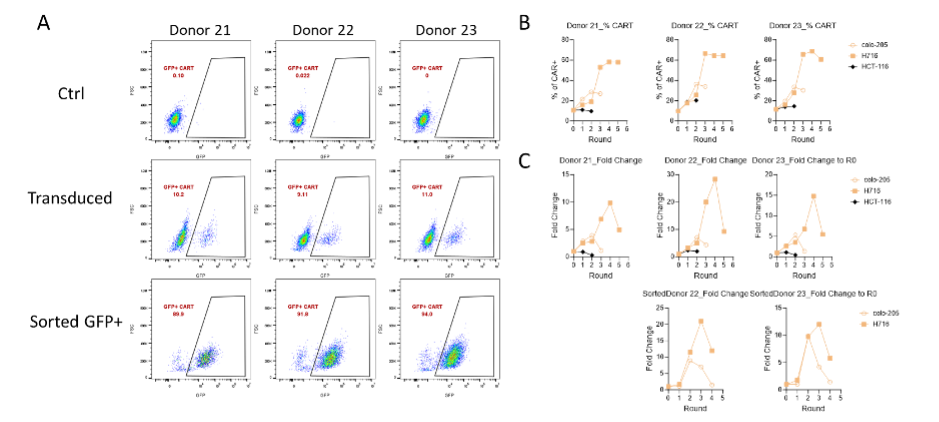
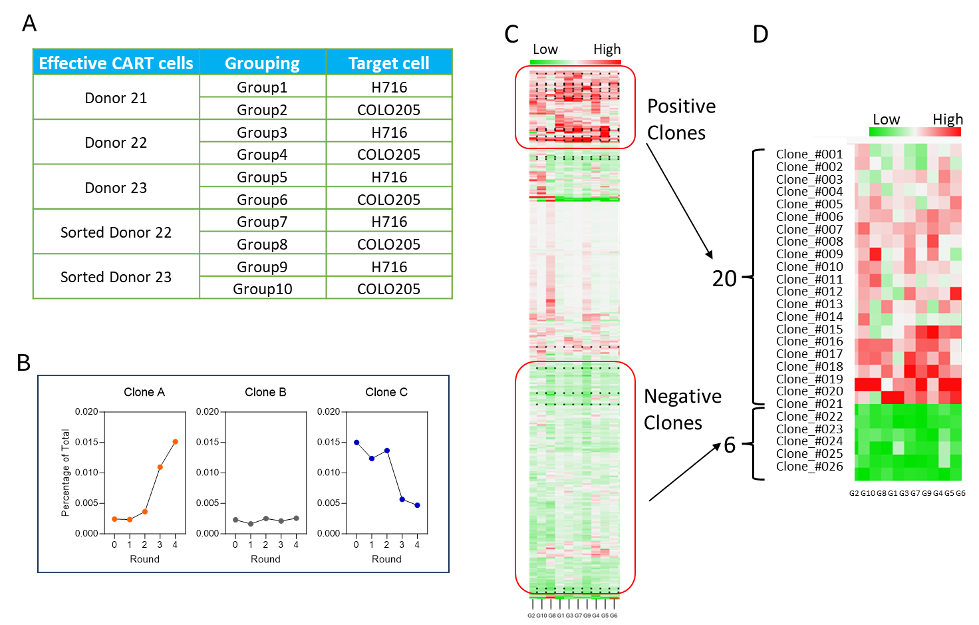 Figure 3. NGS and slope analysis for VHH hit ranking. (A) Grouping information. (B) The proliferation curve shows the clone percentage of each round based on NGS, with the slope of the curve calculated through fitting. Three examples are shown to demonstrate different types of slopes. (C) Hierarchical clustering analysis for lead selection. (D) Twenty positive and six negative clones were selected for validation.
Figure 3. NGS and slope analysis for VHH hit ranking. (A) Grouping information. (B) The proliferation curve shows the clone percentage of each round based on NGS, with the slope of the curve calculated through fitting. Three examples are shown to demonstrate different types of slopes. (C) Hierarchical clustering analysis for lead selection. (D) Twenty positive and six negative clones were selected for validation.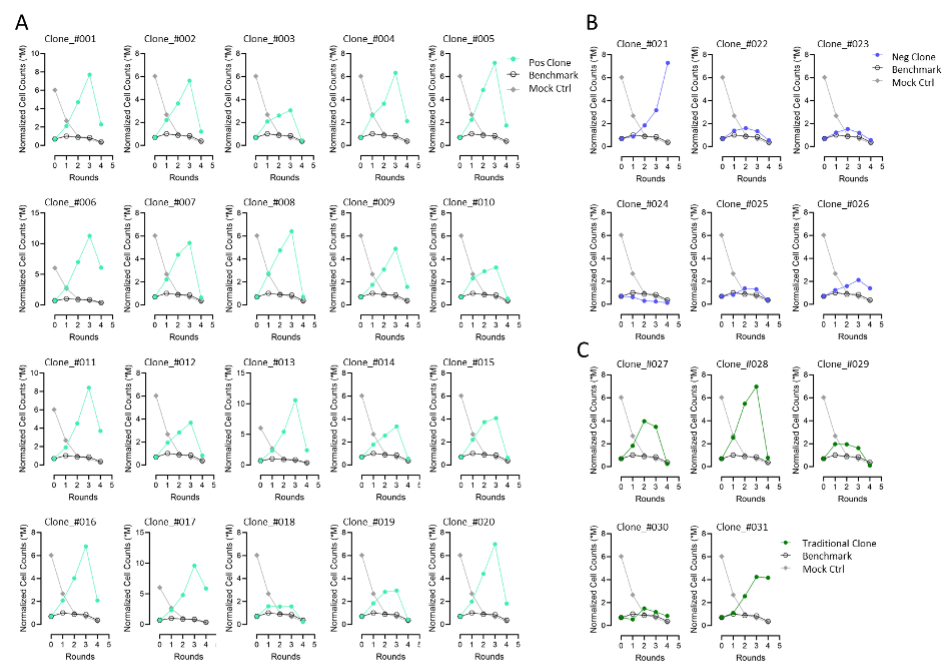
 2024-11-25 IVC & SITC | 上海细胞医治集团纳米抗体装甲化肿瘤疫苗BaizeDC研究进展前后受邀于国际权势巨子学术集会举行分享
2024-11-25 IVC & SITC | 上海细胞医治集团纳米抗体装甲化肿瘤疫苗BaizeDC研究进展前后受邀于国际权势巨子学术集会举行分享  2024-11-23 上海细胞医治集团自立立异创建即时、高疗效、低成本“闪CAR-T”药物平台,得到阶段性临床开发进展,获邀于多个专业平台举行立异分享
2024-11-23 上海细胞医治集团自立立异创建即时、高疗效、低成本“闪CAR-T”药物平台,得到阶段性临床开发进展,获邀于多个专业平台举行立异分享  -米兰官网
-米兰官网